Description
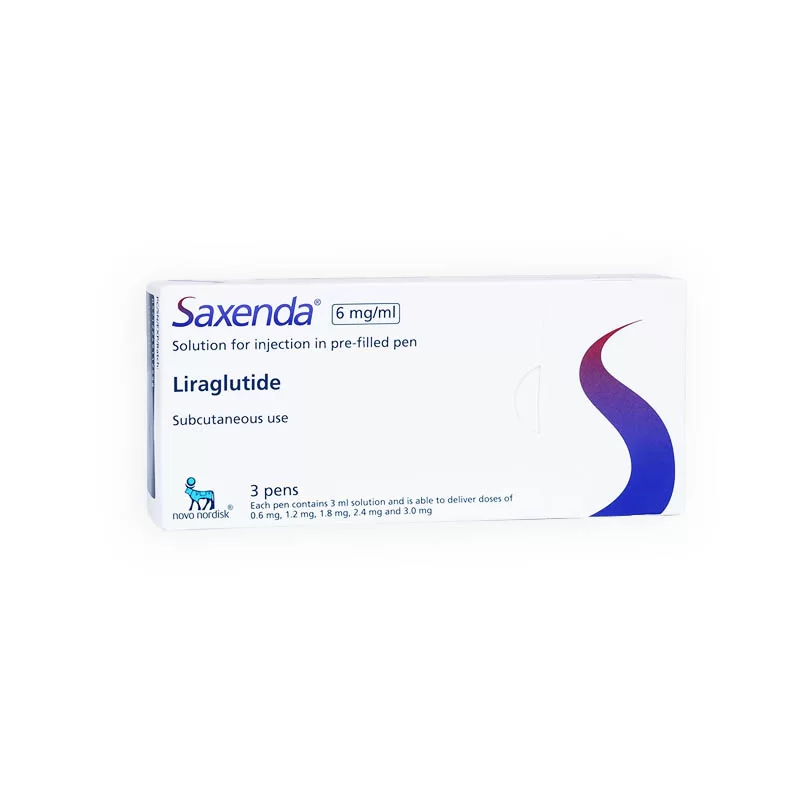
SAXENDA® is indicated for chronic weight management in: Adults with a BMI ≥30 kg/m² (obesity) or ≥27 kg/m² (overweight) with at least one comorbid condition (e.g., type 2 diabetes, hypertension) Adolescents aged 12 years and older with body weight >60 kg and a BMI corresponding to ≥30 kg/m² for adults Contraindications: Personal/family history of medullary thyroid carcinoma (MTC) Presence of Multiple Endocrine Neoplasia syndrome type 2 (MEN 2) Pregnancy or hypersensitivity to liraglutide or excipients Common Side Effects: Nausea (39.3% adults, 42.4% pediatrics) Vomiting, diarrhea, and constipation Injection site reactions , headache , fatigue Rare but serious risks: acute pancreatitis , gallbladder disease , hypoglycemia , suicidal ideation Patients should be assessed after 16 weeks of treatment (adults) or 12 weeks on maintenance dose (pediatrics); therapy should be discontinued if insufficient weight/BMI reduction is observed. SAXENDA® 6mg/ml – 3mg Pen – Administration Technique and Storage SAXENDA® is administered once daily via subcutaneous injection in the abdomen, thigh, or upper arm , independent of meal timing. Dosage is gradually increased to the target 3 mg/day to minimize gastrointestinal side effects. Dose Escalation Schedule: Week 1 : 0.6 mg/day Week 2 : 1.2 mg/day Week 3 : 1.8 mg/day Week 4 : 2.4 mg/day Week 5 onward : 3.0 mg/day Instructions for Use: Rotate injection sites within each region to prevent cutaneous amyloidosis If a dose is missed for >3 days , reinitiate at 0.6 mg/day and restart escalation Do not mix with insulin or other medications in the same injection Storage Guidelines: Unopened pens : Refrigerate at 2°C–8°C After first use : Store at ≤30°C for up to 30 days Do not freeze . Protect from light and keep pen capped Proper training on injection technique and storage is essential for safe and effective SAXENDA® use. Regular monitoring is recommended to ensure sustained weight loss and minimize risk of adverse effects.




Reviews
There are no reviews yet.